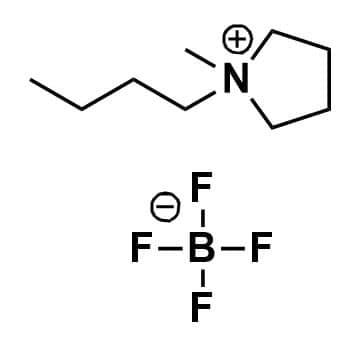
1-Butyl-1-methylpyrrolidinium tetrafluoroborate, >99%
Price range: $178.24 through $6,591.76
Product Code: IL-0077-HPCAS NO: 345984-11-4
- Chemical Formula: C9H20BF4N
- Synonyms: BMPyrr BF4, PYR14 BF4, PY14 BF4, N-Butyl-N-methylpyrrolidinium tetrafluoroborate.
- Anion decomposes slowly in the presence of water.
- Hydrophobic
SUM Formula: C9H20BF4N
Molecular Weight: 229.07
Melting Point: 155 °C
Density: 0.896 g/cm³
Purity: >99%
- SUM Formula: C9H20BF4N
- Molecular Weight: 229.07
- Melting Point: 155 °C
- Density: 0.896 g/cm³
1-Butyl-1-methylpyrrolidinium tetrafluoroborate, CAS: 345984-11-4
Key Applications:
Electrochemical Systems
- Serves as a stable ionic liquid electrolyte component for supercapacitors, electrochemical capacitors, and advanced energy‑storage devices.
- Supports wide electrochemical windows and low volatility, enabling high‑performance operation under demanding conditions.
- Useful as a supporting electrolyte in voltammetric studies, electrodeposition research, and ionic‑liquid‑based redox systems.
Battery and Energy‑Storage Research
- Employed in lithium‑free and lithium‑salt‑containing electrolyte formulations where BF₄⁻ anions provide favorable conductivity and thermal stability.
- Investigated in prototype electrolytes for next‑generation batteries, including dual‑ion and hybrid‑ion systems.
Catalysis and Synthesis
- Functions as a non‑volatile, thermally robust reaction medium for organic transformations requiring controlled ionic environments.
- Applied in phase‑transfer‑like roles where ionic liquids enhance reaction rates, selectivity, or catalyst recyclability.
Materials Science and Thin‑Film Processing
- Utilized in the preparation of ionic‑liquid‑modified polymers, membranes, and composite materials to tune conductivity, mechanical properties, and ion transport.
- Supports electrodeposition of metals and alloys under water‑free conditions, improving film uniformity and morphology.
Separation Science
- Acts as a tunable solvent for extraction of metal ions, organic compounds, and polar analytes, particularly in systems requiring low vapor pressure and high thermal stability.
- Integrated into supported ionic‑liquid membranes (SILMs) for selective gas or solute transport.
Analytical Chemistry
- Serves as a medium for electroanalytical measurements where high ionic strength and chemical inertness are required.
- Used in sample preparation workflows, including ionic‑liquid‑assisted extraction and preconcentration.
Please contact us if you want to learn more or need assistance with your order.