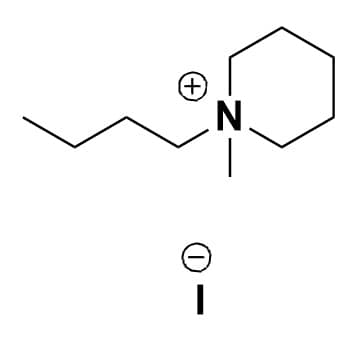
1-Butyl-1-methylpiperidinium iodide, >98%
Price range: $290.11 through $3,237.10
Product Code: IL-0169-HPCAS NO: 37971-78-1
- Chemical Formula: C10H22IN
- Synonyms: N-Butyl-N-methylpiperidinium iodide, BMPip I, PIP14 I
- Weakly-coordinating anion
SUM Formula: C10H22IN
Molecular Weight: 283.19
Melting Point: 192 °C
Density: 1.305 g/cm³ (25 °C)
Purity: >98%
- SUM Formula: C10H22IN
- Molecular Weight: 283.19
- Melting Point: 192 °C
- Density: 1.305 g/cm³ (25 °C)
1-Butyl-1-methylpiperidinium iodide, CAS: 37971-78-1
Key Applications:
Electrochemical Systems
- Serves as a supporting electrolyte in non‑aqueous electrochemical studies where high ionic strength and wide electrochemical windows are required.
- Useful in evaluating ion‑transport behavior in aprotic media due to the bulky, asymmetric piperidinium cation.
- Supports charge‑transfer studies in redox‑active organic and organometallic systems.
Materials Science and Polymer Research
- Incorporated into polymer matrices to study ionic conductivity, plasticization effects, and ion–polymer interactions.
- Enables formulation of ionically conductive films and gels for exploratory solid‑state device architectures.
- Used in structure–property investigations of piperidinium‑based ionic materials.
Phase‑Transfer and Catalytic Environments
- Functions as a phase‑transfer medium in reactions requiring iodide as a nucleophile or halide source.
- Provides a controlled iodide environment for mechanistic studies in substitution, elimination, and metal‑mediated transformations.
- Supports catalytic systems where bulky organic cations improve solubility or stabilize reactive intermediates.
Solvent and Extraction Studies
- Applied in biphasic extraction experiments to probe ion‑pairing, partitioning, and solvation behavior of iodide in organic media.
- Useful for tuning polarity and ionic strength in mixed‑solvent systems for analytical method development.
Spectroscopic and Fundamental Research
- Employed as a model piperidinium iodide salt for NMR, IR, and Raman studies examining cation–anion interactions.
- Supports benchmarking of computational models involving charge distribution, conformational behavior, and ion‑pair energetics.
Please contact us if you want to learn more or need assistance with your order.