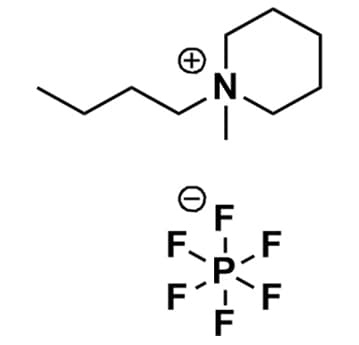
1-Butyl-1-methylpiperidinium hexafluorophosphate, >99%
Price range: $267.49 through $10,393.76
Product Code: IL-0156-HPCAS NO: 1257647-66-7
- Chemical Formula: C10H22F6NP
- Synonyms: N-Butyl-N-methylpiperidinium hexafluorophosphate, BMPip PF6, PIP 14 PF 6
- Anion decomposes slowly in the presence of water
- Weakly-coordinating Anion
- Hydrophobic
SUM Formula: C10H22F6NP
Molecular Weight: 301.25
Melting Point: 81°C
Purity: >99%
- SUM Formula: C10H22F6NP
- Molecular Weight: 301.25
- Melting Point: 81°C
1-Butyl-1-methylpiperidinium hexafluorophosphate, CAS: 1257647-66-7
Key Applications:
Electrochemical Systems
- Serves as a stable, non‑volatile electrolyte component for high‑voltage electrochemical cells.
- Supports wide electrochemical windows, enabling use in advanced capacitor and battery research.
- Useful as an ionic medium for electrodeposition studies requiring low vapor pressure and high thermal stability.
Catalysis and Synthesis
- Functions as a non‑coordinating ionic environment for transition‑metal catalysis, particularly in reactions sensitive to moisture or coordinating solvents.
- Provides a tunable, hydrophobic reaction phase for biphasic catalysis and selective transformations.
- Suitable as a solvent or co‑solvent for nucleophilic substitution, alkylation, and organometallic processes.
Separation Science
- Employed as a stationary‑phase modifier or mobile‑phase additive in chromatography to enhance ion‑pairing and retention control.
- Supports extraction and partitioning studies where hydrophobic ionic liquids improve selectivity and phase behavior.
Materials and Polymer Science
- Acts as a structuring agent or ionic plasticizer in polymer electrolytes and ion‑conductive films.
- Useful in preparing ionogels and hybrid materials requiring thermal stability and low volatility.
- Supports nanoparticle stabilization and templating in ionic‑liquid‑based synthesis.
Spectroscopy and Analytical Chemistry
- Provides a low‑volatility, high‑purity medium for spectroscopic measurements, minimizing background interference.
- Suitable for electroanalytical studies where ionic conductivity and chemical inertness are critical.
General Research Use
- A versatile hydrophobic ionic liquid for fundamental studies in solvation, ion transport, and interfacial behavior.
- Supports method development in fields exploring ionic‑liquid‑based solvents, electrolytes, and reaction media.
Please contact us if you want to learn more or need assistance with your order.