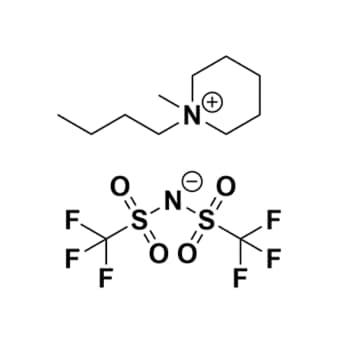
1-Butyl-1-methylpiperidinium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $116.07 through $5,758.00
Product Code: IL-0154-HPCAS NO: 623580-02-9
- Chemical Formula: C12H22F6N2O4S2
- Synonyms: N-Butyl-N-methylpiperidinium bis(trifluoromethylsulfonyl)imide, BMPip BTA, BMPip NTf2, BMPip TFSI, BMPip BTI, PIP14 TFSI
- Weakly-coordination anion
- Hydrophobic
Conductivity: 0.84 mS/cm (25 °C)
SUM Formula: C12H22F6N2O4S2
Molecular Weight: 436.44
Melting Point: -76 °C
Density: 1.38 g/cm³
ECW: 3.7 V
Purity: >99%
Viscosity: 183 cP (20 °C)
- SUM Formula: C12H22F6N2O4S2
- Molecular Weight: 436.44
- Melting Point: -76 °C
- Density: 1.38 g/cm³
- ECW: 3.7 V
- Viscosity: 183 cP (20 °C)
1-Butyl-1-methylpiperidinium bis(trifluoromethylsulfonyl)imide, CAS: 623580-02-9
Key Applications:
Electrochemical Systems
- High‑voltage electrolytes for lithium, sodium, and magnesium batteries, leveraging the wide electrochemical stability window of piperidinium–TFSI salts.
- Ionic liquid electrolytes for supercapacitors and hybrid capacitors requiring low volatility and high thermal stability.
- Electrolyte additives to improve interfacial stability, suppress dendrite formation, and enhance cycling performance in next‑generation metal‑anode systems.
High‑Temperature & Harsh‑Environment Media
- Heat‑resistant ionic liquid matrices for processes operating above 150–200^\circC where conventional organic solvents degrade.
- Stable media for high‑temperature spectroscopy and analytical methods requiring non‑volatile, chemically inert solvents.
- Thermally robust lubricants and anti‑wear additives for extreme‑environment tribology.
Catalysis & Synthesis
- Non‑coordinating reaction media for transition‑metal catalysis, especially where TFSI⁻ enhances catalyst lifetime.
- Solvent or co‑solvent for halide‑sensitive transformations and organometallic reactions.
- Phase‑transfer medium in biphasic systems requiring hydrophobic, non‑nucleophilic ionic liquids.
Separation & Extraction
- Selective extraction of metal ions, particularly hydrophobic complexes of rare‑earth or transition metals.
- Solvent systems for liquid–liquid extraction where low miscibility with water and high chemical stability are advantageous.
- CO₂ capture and gas‑absorption studies, leveraging the tunable viscosity and hydrophobicity of the piperidinium–TFSI framework.
Materials Science & Surface Engineering
- Template or structuring agent in polymer and nanoparticle synthesis, enabling controlled morphology and conductivity.
- Plasticizer or ionic dopant for polymer electrolytes, ion gels, and flexible electronic materials.
- Surface‑modification medium for creating hydrophobic, ion‑conductive coatings.
Industrial & Engineering Uses
- Hydrophobic ionic liquid lubricant base stocks for aerospace, vacuum, and high‑temperature mechanical systems.
- Dielectric fluids in specialized electronics requiring low flammability and high breakdown strength.
- Antistatic and charge‑control additives in polymer processing and advanced manufacturing.
Please contact us if you want to learn more or need assistance with your order.