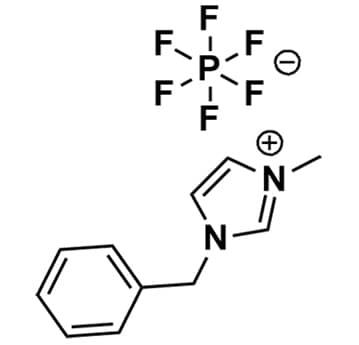
1-Benzyl-3-methylimidazolium hexafluorophosphate, >99%
Price range: $137.20 through $8,587.57
Product Code: IL-0187-HPCAS NO: 433337-11-2
- Chemical Formula:
- Synonyms: BenyMIM PF6
- Anion decomposes slowly in the presence of water
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
SUM Formula: C11H13F6N2P
Molecular Weight: 318.20
Melting Point: 135 °C
Purity: >99%
- SUM Formula: C11H13F6N2P
- Molecular Weight: 318.20
- Melting Point: 135 °C
1-Benzyl-3-methylimidazolium hexafluorophosphate, CAS: 433337-11-2
Key Applications
Solvent and Medium for Organic Synthesis
- Serves as a hydrophobic ionic liquid for transition‑metal‑catalyzed reactions, including cross‑coupling, hydrogenation, and oxidation.
- Enhances reaction rates and selectivity due to strong cation–π interactions from the benzyl substituent.
- Supports biphasic catalysis where product separation is simplified by phase partitioning.
Extraction and Separation Processes
- Effective in liquid–liquid extraction of aromatic compounds, phenolics, and metal ions due to its high affinity for π‑rich substrates.
- Used in selective separation of transition metals and rare‑earth ions when paired with coordinating ligands.
- Suitable for partitioning hydrophobic organics from aqueous matrices.
Electrochemical and Materials Applications
- Functions as a stable electrolyte component in electrochemical cells, benefiting from the PF₆⁻ anion’s wide electrochemical window.
- Employed in electrodeposition studies, ionic‑liquid‑based supercapacitors, and conductive polymer processing.
- Supports formation of ionogels and polymer–ionic liquid composites with tunable conductivity.
Catalysis and Phase‑Transfer Systems
- Acts as a phase‑transfer medium for reactions involving poorly soluble organic substrates.
- Enhances catalytic turnover in systems requiring strong solvation of aromatic intermediates.
- Useful in immobilizing homogeneous catalysts for recycling and reuse.
Spectroscopy and Analytical Chemistry
- Provides a non‑volatile, thermally stable medium for NMR, UV–Vis, and IR studies of aromatic and organometallic species.
- Stabilizes reactive intermediates through π‑cation interactions, enabling mechanistic investigations.
Thermal and Chemical Stability Applications
- Suitable for high‑temperature reaction environments due to the PF₆⁻ anion’s thermal robustness.
- Used in studies requiring low vapor pressure and minimal solvent loss.
Please contact us if you want to learn more or need assistance with your order.