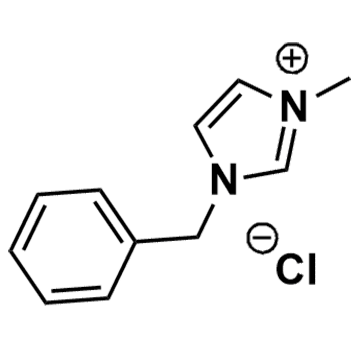
1-Benzyl-3-methylimidazolium chloride, >99%
Price range: $148.58 through $6,188.42
Product Code: IL-0140-HPCAS NO: 36443-80-8
- Chemical Formula: C11H13ClN2
- Synonyms: BenzMIM Cl
- Aromatic
SUM Formula: C11H13ClN2
Molecular Weight: 208.69
Melting Point: 58 °C
Density: 1.673 g/cm³ (25 °C)
Purity: >99%
- SUM Formula: C11H13ClN2
- Molecular Weight: 208.69
- Melting Point: 58 °C
- Density: 1.673 g/cm³ (25 °C)
1-Benzyl-3-methylimidazolium chloride, CAS: 36443-80-8
Key Applications:
Solvent and Reaction Medium
- Serves as a polar, non‑volatile ionic medium for organic synthesis, particularly in transformations benefiting from enhanced cation–π interactions.
- Supports transition‑metal‑catalyzed reactions, including cross‑coupling and C–N/C–C bond‑forming processes, where the benzyl substituent can influence solvation and catalyst stability.
- Useful in phase‑transfer–like environments due to its strong chloride coordination and tunable viscosity.
Catalysis and Organocatalysis
- Functions as a co‑catalyst or catalytic medium in acid‑promoted and nucleophilic substitution reactions.
- Enhances rates of electrophilic aromatic substitutions and benzylation reactions by stabilizing reactive intermediates.
- Employed in supported ionic liquid phase (SILP) catalytic systems, where the aromatic cation improves immobilization on carbonaceous supports.
Materials Science and Polymer Processing
- Acts as a structure‑directing additive in polymerization and copolymerization systems, influencing morphology and ionic conductivity.
- Used in the preparation of polymer electrolytes and ion‑conductive films where the benzyl group contributes to mechanical rigidity and cation mobility.
- Supports dispersion and stabilization of carbon nanomaterials, including CNTs and graphene derivatives, through π–π interactions.
Electrochemical Applications
- Serves as a component in electrolyte formulations for electrochemical devices requiring high ionic strength and chloride availability.
- Applied in electrode surface modification and electrocatalyst preparation, where the aromatic cation can modulate interfacial charge transfer.
Extraction, Separation, and Purification
- Effective in liquid–liquid extraction systems for metal ions and organic solutes, leveraging chloride coordination and aromatic interactions.
- Used in chromatographic stationary phase modification to enhance selectivity for aromatic and polar analytes.
Antimicrobial and Biochemical Research
- Investigated as a cationic antimicrobial agent in microbiological studies, where the benzyl substituent enhances membrane interaction.
- Utilized in protein and enzyme stabilization studies due to its tunable hydrophobicity and ionic strength contribution.
General Laboratory Utility
- Suitable as a precursor for synthesizing other benzyl‑functionalized ionic liquids via anion exchange.
- Employed in formulation studies exploring viscosity, polarity, and solvation effects across imidazolium IL families.
Please contact us if you want to learn more or need assistance with your order.