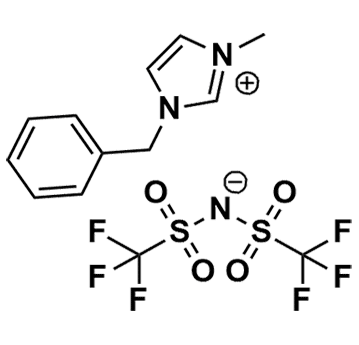
1-Benzyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $168.35 through $9,640.73
Product Code: IL-0241-HPCAS NO: 433337-24-7
- Chemical Formula: C13H13F6N3O4S2
- Synonyms: BenzMIM BTA, BenzMIM NTf 2 , BenzMIM TFSI, BenzMIM BTI
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 0.62 mS/cm (20 °C)
SUM Formula: C13H13F6N3O4S2
Molecular Weight: 453.38
Melting Point: 23 °C
Density: 1.49 g/cm³ (25 °C)
ECW: 4.5V
Purity: >99%
Viscosity: 350 cP (20 °C)
- SUM Formula: C13H13F6N3O4S2
- Molecular Weight: 453.38
- Melting Point: 23 °C
- Density: 1.49 g/cm³ (25 °C)
- ECW: 4.5V
- Viscosity: 350 cP (20 °C)
1-Benzyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 433337-24-7
Key Applications:
Advanced Solvent and Reaction Medium
- Serves as a highly stable, non‑volatile solvent for organic synthesis, particularly where aromatic stabilization or cation–π interactions enhance reactivity.
- Supports transition‑metal catalysis, cross‑coupling, and electrophilic aromatic substitution under mild conditions.
- Enables selective extraction and separation processes due to its hydrophobic, low‑coordinating NTf₂⁻ anion.
Electrochemical and Energy‑Related Uses
- Functions as an ionic liquid electrolyte component with wide electrochemical stability windows.
- Suitable for redox‑active systems where aromatic cation structures improve charge transport or solvation of organic redox couples.
- Used in electrodeposition studies, especially for noble metals and conductive polymer formation.
Materials Science and Polymer Processing
- Acts as a plasticizer or processing aid for specialty polymers, including ionomers and aromatic‑rich matrices.
- Supports templating and structuring in porous materials, carbon frameworks, and ionic‑liquid‑derived composites.
- Useful in stabilizing nanoparticles and dispersions where π‑interactions improve colloidal behavior.
Extraction, Separation, and Purification
- Effective in liquid–liquid extraction of aromatic compounds, metal ions, and hydrophobic organics.
- Applied in biphasic catalysis systems where phase separation and recyclability are essential.
- Offers tunable selectivity for organic contaminants in environmental remediation research.
Spectroscopy, Analytical Chemistry, and Research Applications
- Provides a low‑volatility, high‑purity matrix for NMR, IR, and electroanalytical studies.
- Used to investigate ion pairing, solvation dynamics, and π‑stacking effects in ionic liquid systems.
- Serves as a benchmark aromatic imidazolium IL for structure–property relationship studies.
Please contact us if you want to learn more or need assistance with your order.