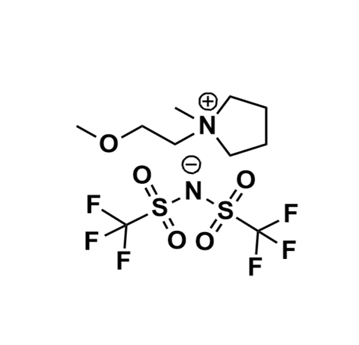
1-(2-Methoxyethyl)-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)imide, >98%
Price range: $69.34 through $1,102.60
Product Code: IL-0359-HPCAS NO: 757240-24-7
- Chemical Formula: C10H18N2O5S2F6
- Synonyms: MeOEtMPyrr BTA
Conductivity: 2,503 mS/cm
SUM Formula: C10H18N2O5S2F6
Molecular Weight: 424.38
Melting Point: -55°C
Density: 1.4449 g/cm³
Purity: >98%
Viscosity: 55.1 cP
- SUM Formula: C10H18N2O5S2F6
- Molecular Weight: 424.38
- Melting Point: -55°C
- Density: 1.4449 g/cm³
- Viscosity: 55.1 cP
1-(2-Methoxyethyl)-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)imide, CAS: 757240-24-7
Key Applications:
Electrochemical Systems
- Lithium metal and lithium‑ion battery electrolytes: Ether-functional cation enhances Li⁺ coordination and interfacial stability; TFSI⁻ provides wide electrochemical windows and high oxidative resistance.
- Supercapacitors and hybrid capacitors: Low viscosity relative to longer-chain pyrrolidinium ILs improves ion mobility and reduces ESR.
- Electrodeposition media: Suitable for controlled deposition of reactive or air‑sensitive metals due to negligible vapor pressure and high thermal stability.
Energy and Thermal Management
- Heat‑transfer and thermal‑storage fluids: High thermal stability and low flammability support use in closed-loop systems requiring non‑volatile, non‑corrosive media.
- Electrothermal actuators and devices: Stable ionic conductivity across broad temperature ranges enables consistent performance in thermally cycled environments.
Catalysis and Synthesis
- Solvent and co‑solvent for transition‑metal catalysis: Ether functionality improves solvation of polar substrates while TFSI⁻ maintains low coordinating behavior.
- Medium for selective organic transformations: Supports reactions requiring high polarity, low nucleophilicity, and thermal robustness.
- Phase‑transfer and biphasic catalysis: Immiscibility with many organic solvents enables catalyst recycling and simplified separation.
Materials Science and Polymer Processing
- Polymer electrolytes and ionogels: Forms stable, conductive networks when immobilized in polymer matrices; ether group enhances miscibility with PEG‑based systems.
- Plasticization and processing aid: Reduces Tg in certain engineering polymers and facilitates melt processing under controlled conditions.
- Nanomaterials synthesis: Provides a stable, tunable environment for nanoparticle nucleation and growth with minimized aggregation.
Analytical and Separation Technologies
- Chromatographic stationary‑phase modifier: Tailors polarity and selectivity in IL‑modified phases.
- Electrophoretic media: High ionic strength and low volatility support stable, reproducible separations.
Surface and Interface Engineering
- Lubrication and anti‑wear films: Strong interfacial adsorption and thermal stability enable use in high‑temperature or vacuum tribological systems.
- Surface functionalization medium: Ether group improves compatibility with oxide and polymer surfaces during modification or grafting processes.
Please contact us if you want to learn more or need assistance with your order.