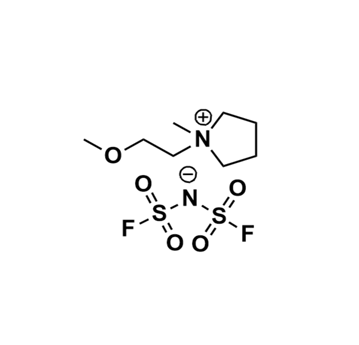
1-(2-Methoxyethyl)-1-methylpyrrolidinium bis(fluorosulfonyl)imide, >99%
Price range: $55.12 through $961.07
Product Code: IL-0360-HPCAS NO: 1235234-47-5
- Chemical Formula: C8H18N2O5S2F2
- Synonyms: MeOEtMPyrr TFI
Conductivity: 4.60 mS/cm
SUM Formula: C8H18N2O5S2F2
Molecular Weight: 324.37
Melting Point: 8°C
Density: 1.4180g/cm³
Purity: >99%
Viscosity: 42.4 cP
- SUM Formula: C8H18N2O5S2F2
- Molecular Weight: 324.37
- Melting Point: 8°C
- Density: 1.4180g/cm³
- Viscosity: 42.4 cP
1-(2-Methoxyethyl)-1-methylpyrrolidinium bis(fluorosulfonyl)imide, CAS: 1235234-47-5
Key Applications:
Advanced Electrolytes
- Serves as a high‑conductivity ionic liquid electrolyte component for lithium, sodium, and multivalent metal batteries.
- Enhances low‑temperature ionic mobility due to the flexible 2‑methoxyethyl side chain.
- Supports high‑voltage cathode chemistries when paired with FSI⁻, which offers fast anion transport and favorable SEI formation.
Capacitors and Energy Storage
- Used in electrochemical double‑layer capacitors (EDLCs) and hybrid capacitors requiring low viscosity and high ionic conductivity.
- Improves cycling stability in devices operating across wide temperature ranges.
Electrodeposition and Metal Processing
- Functions as a non‑volatile, thermally stable medium for controlled metal deposition, especially for reactive metals where traditional solvents are unsuitable.
- Enables smooth, uniform metal films due to the FSI⁻ anion’s favorable coordination behavior.
Organic Synthesis and Catalysis
- Acts as a tunable, polar, aprotic solvent for transition‑metal‑catalyzed reactions.
- Supports high‑selectivity transformations where moisture‑free, thermally robust media are required.
Separation Science
- Utilized in liquid–liquid extraction systems for selective metal ion separation, particularly for rare‑earth and transition‑metal purification.
- Offers improved phase behavior and solvation characteristics due to the ether‑functionalized cation.
Thermal and Chemical Stability Applications
- Suitable for high‑temperature environments where conventional organic solvents degrade.
- Employed in specialty formulations requiring low vapor pressure and strong electrochemical stability.