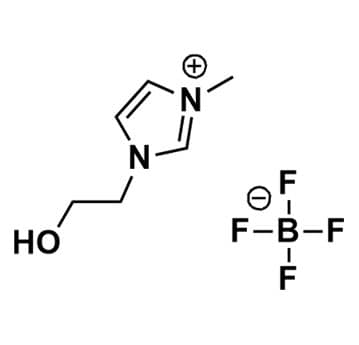
1-(2-Hydroxyethyl)-3-methylimidazolium tetrafluoroborate, >98%
Price range: $128.80 through $1,163.41
Product Code: IL-0038-HPCAS NO: 374564-83-7
- Chemical Formula: C6H11BF4N2O
- Synonyms: HO-EMIM BF4
- Anion decomposes slowly in the presence of water
- Hydrophilic
- Aromatic
Conductivity: 3.42 mS/cm (20 °C)
SUM Formula: C6H11BF4N2O
Molecular Weight: 213.97
Melting Point: -44 °C
Density: 1.34 g/cm³ (22 °C)
Purity: >98%
Viscosity: 137 cP (20 °C)
- SPECIFIC GRAVITY: NA
- SUM Formula: C6H11BF4N2O
- Molecular Weight: 213.97
- Melting Point: -44 °C
- Density: 1.34 g/cm³ (22 °C)
- TSCA: NA
- Viscosity: 137 cP (20 °C)
1- (2-Hydroxyethyl)-3-methylimidazolium Tetrafluoroborate, CAS: 374564-83-7
Key Applications:
1. Solvent and Co‑solvent in Organic Synthesis
- Supports acid‑catalyzed and metal‑catalyzed transformations due to its polar, mildly coordinating environment.
- Hydroxyl group enhances miscibility with water and alcohols, enabling biphasic or monophasic reaction tuning.
- Useful in nucleophilic substitution, esterification, and condensation reactions where controlled polarity is beneficial.
2. Medium for Biocatalysis
- Hydroxyl-functionalized ionic liquids often improve enzyme stability and activity relative to non-functionalized ILs.
- Suitable for lipase‑catalyzed esterifications, transesterifications, and kinetic resolutions.
- BF₄⁻ salts with hydroxyethyl cations can modulate water activity, improving selectivity in biotransformations.
3. Electrochemical Applications
- Serves as a conductive electrolyte or electrolyte additive in systems requiring high ionic strength and low volatility.
- Hydroxyl group increases hydrogen‑bonding networks, influencing viscosity and ionic mobility.
- Applicable in supercapacitors, electrochemical sensors, and electrodeposition environments where protic ILs improve interface behavior.
4. Gas Absorption and Separation
- Hydroxyethyl-functionalized ILs show enhanced CO₂ solubility via physical absorption and weak chemisorption.
- Can be used in CO₂ capture, acid gas scrubbing, and gas–liquid separation studies.
- BF₄⁻ anion provides relatively low viscosity compared to more strongly coordinating anions, improving mass transfer.
5. Catalytic Support and Reaction Medium for Green Chemistry
- Acts as a recyclable reaction medium for catalytic systems (e.g., Pd, Cu, Brønsted acids).
- Hydroxyl group can participate in secondary interactions that stabilize catalytic intermediates.
- Useful in microwave‑assisted and solvent‑free reaction protocols.
6. Polymer and Material Processing
- Functions as a plasticizer or processing aid for polar polymers such as PVA, cellulose derivatives, and polyamides.
- Hydroxyl-functionalized ILs can dissolve or swell biopolymers, enabling film formation, fiber spinning, or composite preparation.
- BF₄⁻ salts are often used in ion-conductive polymer blends.
7. Extraction and Separation Processes
- Effective in liquid–liquid extraction of metal ions, dyes, and polar organics due to tunable polarity.
- Hydroxyl group enhances selectivity for hydrogen‑bonding solutes.
- Useful in aqueous biphasic systems (ABS) when paired with salts or polymers.
8. Thermal Fluids and Heat‑Transfer Media
- High thermal stability and negligible vapor pressure make it suitable for heat‑transfer and thermal storage studies.
- Hydroxyl functionality can modulate heat capacity and viscosity relative to alkyl-only ILs.
Please contact us if you want to learn more or need assistance with your order.