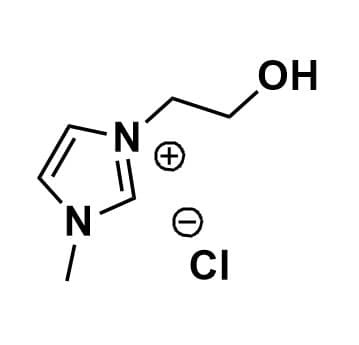
1-(2-Hydroxyethyl)-3-methylimidazolium chloride, >99%
Price range: $107.54 through $3,497.54
Product Code: IL-0039-HPCAS NO: 61755-34-8
- Chemical Formula: C6H11ClN2O
- Synonyms: HO-EMIM Cl
- Hydrophilic
- Aromatic
SUM Formula: C6H11ClN2O
Molecular Weight: 162.62
Melting Point: 83 °C
Density: 1.300 g/cm³ (26 °C)
Purity: >99%
- SPECIFIC GRAVITY: NA
- SUM Formula: C6H11ClN2O
- Molecular Weight: 162.62
- Melting Point: 83 °C
- Density: 1.300 g/cm³ (26 °C)
- TSCA: NA
1-(2-Hydroxyethyl)-3-methylimidazolium chloride, CAS: 61755-34-8
Key Applications:
1. Solvent and Co‑solvent in Catalysis
- Enhances solubility of polar substrates and metal catalysts.
- Hydroxyl group improves coordination with transition‑metal centers, supporting homogeneous catalysis.
- Used in acid‑catalyzed esterifications, dehydration reactions, and biomass‑derived platform molecule upgrading.
2. Biomass Processing and Carbohydrate Chemistry
- Effective for dissolving cellulose, hemicellulose, and lignin fragments due to strong hydrogen‑bonding capability.
- Supports pretreatment of lignocellulosic feedstocks for biofuel and biochemical production.
- Facilitates selective hydrolysis and derivatization of polysaccharides.
3. Electrolytes for Electrochemical Systems
- High ionic conductivity and strong hydrophilicity make it suitable for aqueous and mixed‑solvent electrolytes.
- Applied in supercapacitors, electrochemical sensors, and electrodeposition baths.
- Chloride anion supports metal‑ion complexation in electrodeposition of Cu, Ni, and Co.
4. Extraction and Separation Processes
- Used in aqueous biphasic systems (ABS) for protein, enzyme, and metal‑ion partitioning.
- Hydroxyl functionality improves phase behavior and selectivity in ABS formulations.
- Supports separation of rare‑earth ions and transition metals in hydrometallurgical workflows.
5. Stabilization of Enzymes and Biocatalysis
- Enhances enzyme solubility and stability in nontraditional media.
- Applied in lipase‑catalyzed esterifications, oxidations, and kinetic resolutions.
- Hydroxyl group reduces denaturing effects compared to more hydrophobic imidazolium salts.
6. Polymer Synthesis and Processing
- Acts as a reaction medium for ring‑opening polymerization and controlled radical polymerization.
- Useful as a plasticizer or ionic additive in hydrophilic polymer matrices (e.g., PVA, PEG).
- Supports fabrication of ion‑conducting polymer films and hydrogels.
7. Corrosion Inhibition
- Adsorbs strongly onto steel and copper surfaces through imidazolium ring and hydroxyl group.
- Demonstrates inhibition performance in acidic chloride environments.
- Used in pickling, pipeline protection, and industrial water systems.
8. CO₂ Capture and Gas Absorption
- Hydroxyl group enhances chemisorption and physisorption interactions with CO₂.
- Applied in aqueous IL blends for post‑combustion capture and gas‑scrubbing research.
9. Nanomaterials and Surface Modification
- Serves as a templating or stabilizing agent for nanoparticles (Au, Ag, Fe₃O₄).
- Supports controlled growth of nanostructures due to hydrogen‑bonding and ionic interactions.
- Used in surface functionalization of silica, graphene oxide, and metal oxides.
10. Organic Synthesis Medium
- Promotes SN1/SN2 reactions, cyclizations, and nucleophilic substitutions.
- Hydroxyl functionality modulates polarity and reduces volatility, enabling greener reaction conditions.
Please contact us if you want to learn more or need assistance with your order.