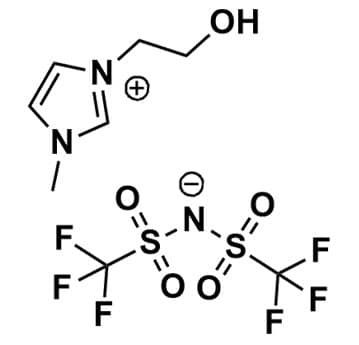
1-(2-Hydroxyethyl)-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $142.89 through $7,818.96
Product Code: IL-0194-HPCAS NO: 174899-86-6
- Chemical Formula: C8H11F6N3O5S2
- Synonyms: HOEMIM BTA, HOEMIM NTf2, HOEMIM TFSI, HOEMIM BTI, hydroxyEMIM BTA, Im1,2O1 BTA
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 1.68 mS/cm (25 °C)
SUM Formula: C8H11F6N3O5S2
Molecular Weight: 407.31
Melting Point: -41 °C
Density: NA
ECW: NA
HMIS Key: NA
Purity: >99%
Viscosity: 81.6 cP (25 °C)
- SPECIFIC GRAVITY: NA
- SUM Formula: C8H11F6N3O5S2
- Molecular Weight: 407.31
- Melting Point: -41 °C
- Density: NA
- ECW: NA
- HMIS KEY: NA
- TSCA: NA
- Viscosity: 81.6 cP (25 °C)
1-(2-Hydroxyethyl)-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 174899-86-6
Key Applications:
Electrochemical Systems
- Serves as a high‑stability ionic liquid electrolyte for lithium, sodium, and multivalent ion batteries, leveraging the TFSI anion’s wide electrochemical window and the cation’s enhanced polarity.
- Used in supercapacitors and hybrid capacitors where low volatility and high ionic conductivity are required.
- Supports electrodeposition studies of metals and alloys under water‑free, thermally stable conditions.
Catalysis and Synthesis
- Functions as a polar, non‑volatile reaction medium for acid‑catalyzed, transition‑metal‑catalyzed, and biocatalytic transformations.
- The hydroxyethyl substituent enables improved solvation of polar substrates and intermediates, supporting selective transformations and rate enhancements.
- Applied in biphasic catalysis where phase separation and ionic liquid recyclability are advantageous.
Separation Science
- Effective as an extraction phase for metal ions, organic acids, and polar organics due to its tunable polarity and strong hydrogen‑bonding capability.
- Utilized in liquid–liquid extraction systems requiring low miscibility with nonpolar solvents and high thermal stability.
- Supports chromatographic method development as a stationary‑phase additive or mobile‑phase modifier.
Materials and Polymer Science
- Incorporated into polymer electrolytes and ion‑conducting membranes to enhance ionic mobility, mechanical flexibility, and thermal stability.
- Used as a plasticizer or conductivity enhancer in solid polymer matrices, including PEO‑based and imidazolium‑functionalized polymers.
- Enables fabrication of ionogels with stable mechanical properties and broad electrochemical windows.
Surface and Interface Engineering
- Applied in surface modification protocols where the hydroxyethyl group promotes adhesion to oxide surfaces.
- Supports formation of stable interfacial layers in electrochemical devices, improving charge transfer and reducing degradation.
- Used in lubrication studies as a low‑volatility, thermally robust ionic medium.
CO₂ Capture and Gas Handling
- Demonstrates strong CO₂ solubility and selectivity, making it suitable for absorption, separation, and catalytic conversion studies.
- The hydroxyl functionality enhances interactions with acidic gases and supports reversible sorption processes.
Analytical and Spectroscopic Applications
- Serves as a stable, low‑vapor‑pressure solvent for NMR, IR, and electrochemical spectroscopy.
- Useful in studying solvation dynamics, ion pairing, and hydrogen‑bonding networks due to its well‑defined structure and high purity.
Contact us if you want to learn more or need assistance with your order.